Daijiworld Media Network - Denver
Denver, Apr 1: Treatment with Remibrutinib has demonstrated rapid and early symptom control in patients with Chronic Spontaneous Urticaria (CSU), according to new post-hoc analysis presented at the American Academy of Dermatology Annual Meeting 2026 in Denver.
CSU is a long-term condition marked by recurring hives, itching, and swelling lasting more than six weeks. It disproportionately affects women, particularly those aged between 20 and 40, and is associated with significant psychological distress and economic burden, with annual healthcare costs in the US exceeding $200 million.
Remibrutinib, an oral and highly selective Bruton's tyrosine kinase inhibitor, had previously shown promising results in earlier trials. In the phase 3 REMIX-1 and REMIX-2 studies, it was administered as an add-on therapy for patients whose symptoms persisted despite standard antihistamine treatment.
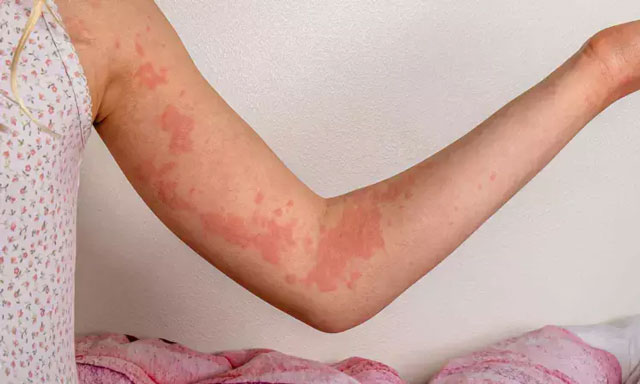
The latest pooled analysis revealed that patients receiving remibrutinib experienced noticeable reductions in hive severity and itching within just 12 hours of the first dose—significantly faster than those given a placebo. A higher proportion of patients also achieved complete symptom resolution, defined as no hives and no itching, within the same timeframe.
The benefits were sustained over the first week of treatment, with patients on remibrutinib consistently showing better symptom control compared to the placebo group.
Safety findings remained consistent with previous studies, with the drug being generally well-tolerated and no new concerns identified.
Researchers noted that the drug’s rapid onset of action could be particularly beneficial for CSU patients who continue to suffer despite existing treatments, reinforcing its potential as an effective therapeutic option.